ChemicalBook > Articles Catagory List >Catalyst-and-Auxiliary >what-is-di-chlorobis-1-2-3-1-phenyl-2-propenyl-dipalladium-ii-
What is di-chlorobis[(1,2,3-)-1-phenyl-2-propenyl]dipalladium(ii)?
Feb 12,2020
Di-chlorobis[(1,2,3-)-1-phenyl-2-propenyl]dipalladium(ii) is an important organic regant for the use of transition-metal-mediated organic syntheses. Di-chlorobis[(1,2,3-)-1-phenyl-2-propenyl]dipalladium(ii) can be used as a catalyst for the ammonia cross-coupling reactions to synthesize arylamines and conversion of aryl triflates to fluorides.
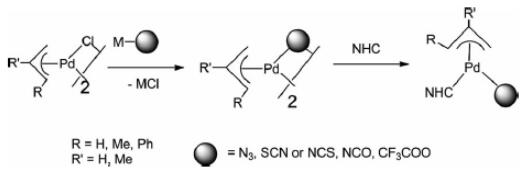
In addition, di-chlorobis[(1,2,3-)-1-phenyl-2-propenyl]dipalladium(ii) is an important organic intermediate to synthesize (π-Allyl)Pd complexes containing N-heterocyclic carbene and pseudohalogen ligands. In transition-metal-mediated organic syntheses, Pd-catalyzed C–C cross-coupling reactions are widely utilized. In particular, palladium compounds containing labile ligands such as C,N-donor ligands have emerged as efficient precatalysts for Suzuki–Miyaura cross-coupling reactions. Furthermore, to enhance catalytic efficiency in the catalytic reactions, several studies on cyclopalladated compounds containing N-heterocyclic carbenes (NHCs), one of the many sterically and/or electronically beneficial ligands, were used. Pseudohalogen ligands are known to be better leaving groups than halogen ligands in catalytic reactions. Their catalytic activity was dependent on the supporting ligand, a tertiary phosphane or an NHC ligand. For example, compounds with an NHC ligand showed higher catalytic activity for aryl chlorides than those with a combinationof C,N-donor and tertiary phosphane ligands [1].
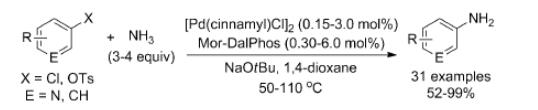
In using [{Pd (cinnamyl)Cl}2]/Mor-DalPhos precatalyst mixtures, electron-rich, electron-poor, and heterocyclic products of ammonia monoarylation were prepared (50–110OC) in high isolated yields. Mor-DalPhos has also been found to be useful in facilitating the Pd-catalyzed monoarylation of hydrazine, acetone, and other carbonyl compounds in chemoselective and aqueous BHA chemistry, as well as in the Au-catalyzed hydroamination of internal alkynes with dialkylamines. Whereas the room-temperature BHA of aryl chlorides with ammonia was not successful when using [{Pd (cinnamyl)Cl}2]/Mor-DalPhos precatalyst mixtures, unprecedented room-temperature transformations of a small number of aryl chlorides were achieved with good monoarylation selectivity by use of the chlorobenzene oxidative addition product [(k2-P,N-Mor-Dal-Phos)Pd(Ph)Cl] as a precatalyst. It is possible to overcome the limitations of other catalysts which include substrate-scope challenges with respect to the monoarylation of electronically deactivated and sterically unhindered aryl chloride substrates, the frequent requirement of relatively high reaction temperatures.[2]
References
1.Kim HK et al. (π-Allyl)Pd Complexes Containing N-Heterocyclic Carbene and Pseudohalogen Ligands – Synthesis, Reactivity toward Organic Isothiocyanates and Isocyanides, and Their Catalytic Activity in Suzuki–Miyaura Cross-Couplings[J]. Eur. J. Inorg. Chem. 2013, 4958–4969
2. Alsabeh PG et al. An Examination of the Palladium/Mor-DalPhos Catalyst System in the Context of Selective Ammonia Monoarylation at Room Temperature[J]. Chem. Eur. J. 2013, 19, 2131 – 2141.
);
You may like
Polyurethane synthesis and Foamed polyurethanes
Jul 26, 2024
polypropylene vs polycarbonate
Mar 18, 2024
Related articles And Qustion
See also
Formic Acid
Apr 30, 2019
Lastest Price from Di-chlorobis[(1,2,3-)-1-phenyl-2-propenyl]dipalladium(II) manufacturers
Di-chlorobis[(1,2,3-)-1-phenyl-2-propenyl]dipalladium(II)
![12131-44-1 Di-chlorobis[(1,2,3-)-1-phenyl-2-propenyl]dipalladium(II)](/ProductImageEN/2023-01/Small/e5013789-c6e1-45e5-9ca4-f703de0175e8.jpg)
US $0.00/KG2024-09-18
- CAS:
- 12131-44-1
- Min. Order:
- 1KG
- Purity:
- 98%min
- Supply Ability:
- 30tons/month
Di-chlorobis[(1,2,3-)-1-phenyl-2-propenyl]dipalladium(II)
![12131-44-1 Di-chlorobis[(1,2,3-)-1-phenyl-2-propenyl]dipalladium(II)](/ProductImageEN/2022-09/Small/c18dac70-4adf-4b70-8713-082516f7970b.png)
US $0.00/kg2022-09-24
- CAS:
- 12131-44-1
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 1000kg
![12131-44-1 di-chlorobis[(1,2,3-)-1-phenyl-2-propenyl]dipalladium(ii); catalyst; reaction; application](https://www.chemicalbook.com/CAS/20211123/GIF/12131-44-1.gif)