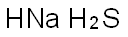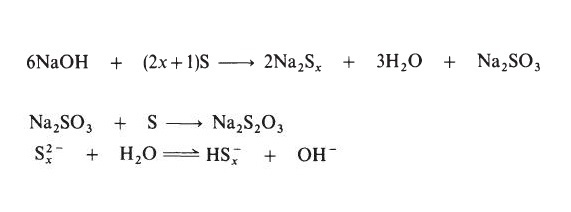Chemical Properties
Yellow-brown, granular, free-flowing
polymer.Combustible.
Chemical Properties
Sodium polysulphides have the general formula Na2Sx where x can be 2-6.
All of these compounds have been prepared in the solid state, in which they
are well defined.
Uses
The sodium polysulphide solutions used for the preparation of polysulphide
elastomers generally have a rank of appro'ximately 2; for elastomers
with a high sulphur content which have especially good solvent resistance, a
rank of about 4 is used. In general, the rank of a polymer is close to that of the
sodium polysulphide solution from which it is formed.
Uses
Manufacture of sulfur dyes and colors, insec-
ticides, oil-resistant synthetic rubber (“Thiokol”
[Toray]), petroleum additives, electroplating.
Preparation
Sodium polysulphide solutions are most commonly prepared by treating
sulphur with aqueous sodium hydroxide:
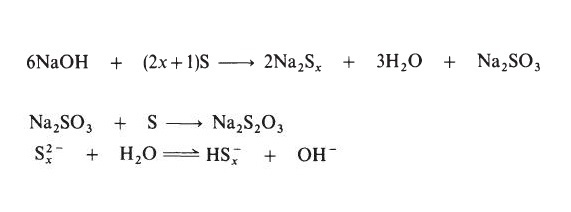
Flammability and Explosibility
Non flammable