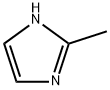
Chemical Properties
2-Methylimidazole is a white to light yellow crystalline powder with an amine-like odor. It is highly soluble in polar organic solvents. Insoluble in ether and cold benzene, easily soluble in water, alcohols and ketones. Irritating to skin and mucous membranes. Mice orally LD50 1400mg/kg.
Chemical Properties
The 2-methylimidazole molecule is approximately planar, and the maximum deviation of all non-H atoms from the plane of the least squares imidazole is 0.006(2)?. N–H··· N hydrogen bonds link molecules together to form infinite chains of hydrogen bond pattern C(4). The crystal of 2-methylimidazole is in orthorhombic crystal system with space group P2
12
12
1 (Z = 4), lattice parameters: a = 5.9957(12) ?, b = 8.1574(16) ? and c = 9.7010(19) ?, V = 474.47(16) ?[1].
Uses
2-Methylimidazole is a monomethylated imidazole that can be used as a building block in the preparation of a wide range of biologically active compounds. 2-Methylimidazole as well as other imidazoles
can be use as catalyst for refolding of enhanced coloured fluorescent protein. 2-Methylimidazole has been identified as a byproduct of fermentation and is detected in foods and mainstream and side-str
eam tobacco smoke.
Uses
2-Methylimidazole is used as a raw material for the preparation of nitroimidazole antibiotics, which is useful in combat anaerobic bacterial and parasitic infections. It acts as a ligand in coordination chemistry. It is also employed as a hardener or accelerator for epoxy resin and auxiliary agent for textile dyes. Further, it acts as a catalyst for refolding of enhanced colored fluorescent protein. It is also useful as a building block in the synthesis of biologically active compounds. In addition to this, it is used as an active pharmaceutical ingredient intermediate such as metronidazole and dimetridazole.
Preparation
2-Methylimidazole is prepared by condensation of glyoxal, ammonia and acetaldehyde, a Radziszewski reaction. It is widely used as a polymeriza tion cross-linking accelerator and a hardener for epoxy resin systems for semiconductor potting compounds and soldering masks.
It is obtained by eliminating dehydrogenation of 2-methylimidazoline. 2-methylimidazoline heated to melt (melting point 107 ℃), carefully add active nickel, raise the temperature to 200-210 ℃ reaction 2h. cool down to below 150 ℃, add water to dissolve, while hot pressure filtration, separation of active nickel, the filter Chemicalbook liquid concentrated to a temperature of 140 ℃ or more, put the material cooling that 2-methylimidazole. Use the method to produce purity of ≥ 98% of the product, 1t product consumption of ethylenediamine (95%) 1095kg, acetonitrile 975kg. better method is to use glyoxal and aldehyde as raw materials.
General Description
It is a white or colorless solid that is highly soluble in polar organic solvents and water. It is a precursor to a range of drugs and is a ligand in coordination chemistry.
Hazard
Possible carcinogen.
Flammability and Explosibility
Not classified
Purification Methods
Recrystallise 2-methylimidazole from *benzene or pet ether. The picrate has m 215o (from H2O). [Beilstein 23 III/IV 594, 23/5 V 35.]
References
[1] Hachu?a B, et al. Crystal and Molecular Structure Analysis of 2-Methylimidazole. Journal of Chemical Crystallography, 2009; 40: 201–206.