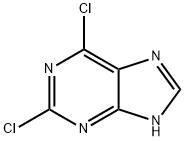
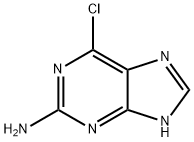
Chemical Properties
white to light yellow crystal powder
Uses
Suzuki-Miyaura cross-coupling between halopurines and arylboronic acids in water-acetonitrile.1
Uses
2,6-Dichloropurine is used in the synthesis of 2,6-diamino-substituted purine derivatives as potential cardiomyogenesis inducing agents.
Synthesis Reference(s)
Journal of the American Chemical Society, 80, p. 404, 1958
DOI: 10.1021/ja01535a040
Purification Methods
It can be recrystallised from 150 parts of boiling H2O and dried at 100o to constant weight. It is soluble in EtOAc. The HgCl2 salt separates from EtOH solution. UV: max 275nm ( 8.9K) at pH 1; and 280nm ( 8.5K) at pH 11 [Elion & Hitchings J Am Chem Soc 78 3508 1956, Schaeffer & Thomas J Am Chem Soc 80 3738 1958, Beaman & Robins J Appl Chem (London) 12 432 1962, Montgomery J Am Chem Soc 78 1928 1956]. [Beilstein 26 III/IV 1747.]