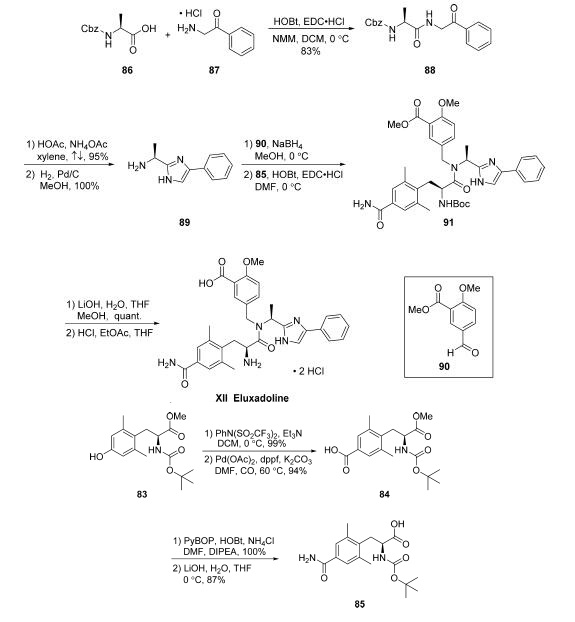
The synthesis of eluxadoline begins with preparation of
advanced coupling component 85, which could be completed
via a four-step route from commercially available N-Bocprotected
aminoester 83 . Triflate formation using N-phenyltrifluoromethanesulfinimide in DCM under
basic conditions led to nearly quantitative yield of the desired
triflate, which was subjected to a carbonylation reaction to yield
aryl acid 84 in 94% yield. Employing NH4Cl as a source of
ammonia, amidation of 84 took place in the presence of
PyBOP/HOBt and DIPEA in DMF. Finally, acid 85 was
revealed upon methyl ester saponification with aqueous LiOH
in THF. This sequence provided 85 without purification.
With coupling component 85 in hand and initiated from a HOBt and EDC?¤HCl-mediated coupling of commercial
N-Cbz-L-alanine (86) with commercial 2-amino acetophenone
hydrochloride (87) to provide intermediate 88 in 83%
yield. Addition of NH4OAc and AcOH to a suspension
of 88 in refluxing xylenes furnished the desired imidazole in
excellent yield (95%). Submission of this N-Cbz-imidazole to
hydrogenation conditions (H2, Pd/C, MeOH) enabled
liberation of the free amine to access 89 in quantitative yield
following filtration and concentration. From intermediate 89,
reductive amination with commercially available aryl aldehyde
90 under standard conditions (NaBH4, MeOH) followed by
subsequent coupling of the corresponding crude amine with acid 85 using HOBt/EDC?¤HCl enabled formation of the
carbon framework of eluxadoline (91). Saponification of the
ester within 91 with LiOH in MeOH/THF yielded the
corresponding acid in quantitative yield. Immediate subjection
of this intermediate to acidic conditions (HCl in EtOAc/THF)
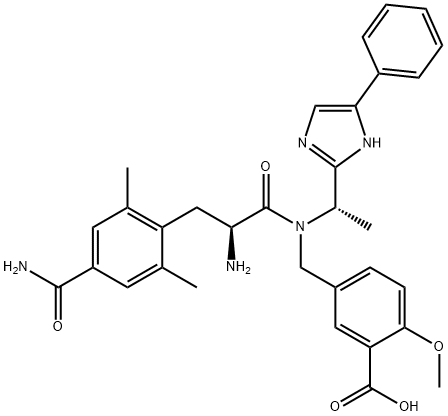
led to N-Boc cleavage and isolation of eluxadoline (XII) as the
bis-HCl salt in 71% yield, requiring no further purification.
It should be noted that since this initial report, additional
details for the isolation of eluxadoline in high purity in various
crystal forms and as a zwitterion have been reported,66 although
most reported routes described isolation of this drug in its HCl
salt form.