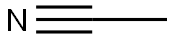General Description
A colorless limpid liquid with an aromatic odor. Flash point 42°F. Density 0.783 c/cm3. Toxic by skin absorption. Less dense than water. Vapors are denser than air.
Reactivity Profile
ACETONITRILE(75-05-8) decomposes when heated to produce deadly toxic hydrogen cyanide gas and oxides of nitrogen. Strongly reactive [Hawley]. May react vigorously with strong oxidizing reagents, sulfuric acid, chlorosulfonic acid, sulfur trioxide, perchlorates, nitrating reagents, and nitric acid. [Sax, 9th ed., 1996, p. 20]. Potentially explosive in contact with nitrogen-fluorine compounds (e.g., tetrafluorourea) [Fraser, G. W. et al., Chem. Comm., 1966, p. 532].
Air & Water Reactions
Highly flammable. Water soluble.
Health Hazard
Acetonitrile liquid or vapor is irritating to the skin, eyes, and respiratory tract. Acetonitrile
has only a modest toxicity, but it can be metabolized in the body to hydrogen cyanide
and thiocyanate. Acetonitrile causes delayed symptoms of poisoning (several hours after
the exposure) that include, but are not limited to, salivation, nausea, vomiting, anxiety,
confusion, hyperpnea, dyspnea, respiratory distress, disturbed pulse rate, unconscious-
ness, convulsions, and coma. Cases of acetonitrile poisoning in humans (or, more strictly,
of cyanide poisoning after exposure to acetonitrile) are rare but not unknown, by inha-
lation, ingestion, and (possibly) by skin absorption. Repeated exposure to acetonitrile
may cause headache, anorexia, dizziness, weakness, and macular, papular, or vesicular
dermatitis.
Health Hazard
Exposure to 160 ppm for 4 hours causes flushing of the face and a feeling of constriction in the chest; 500 ppm for brief periods is irritating to the nose and throat. Severe exposures cause irritability, skin eruptions, confusion, delirium, convulsions, paralysis, and death due to central nervous system depression.
Potential Exposure
Acetonitrile is used as an extractant for animal and vegetable oils, as a solvent; particularly in the pharmaceutical industry, and as a chemical intermediate in pesticide manufacture; making batteries and rubber products. It is present in cigarette smoke
First aid
Acetonitrile can cause fatal Cyanide poisoning. A cyanide antidote kit should be kept in the immediate work area and must be rapidly available. Kit ingredients should be replaced every 1 2 years to ensure freshness. Persons trained in the use of this kit; oxygen use, and CPR must be available within 1 2 minutes. In the event of overexposure, and/or symptoms: move victim to fresh air; call emergency medical care. Give Amyl Nitrate capsules (as directed, by trained personnel only). If this chemical gets into the eyes, remove any contact lenses at once, and irrigate immediately for at least 15 minutes, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts the skin, quickly remove contaminated clothing and wash with large amounts of water. Speed in removing material from skin is of extreme importance. Seek medical attention immediately. When this chemical has been swallowed, get medical attention immediately. If this chemical has been inhaled, remove from exposure and transfer promptly to a medical facility. If not breathing, give artificial respiration (avoid mouth to mouth resuscitation). If breathing is difficult, give oxygen. If heart has stopped, begin CPR. Keep victim quiet and maintain normal body temperature. Effects may be delayed; keep victim under observation. Avoid contact with contaminated skin. Observe victim for 24 to 48 hours.
Shipping
UN1648 Acetonitrile, Hazard Class: 3; Labels: 3-Flammable liquid
Incompatibilities
Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, chlorosulfonic acid, oleum, epoxides. May accumulate static electrical charges, and may cause ignition of its vapors. Nitriles may polymerize in the presence of metals and some metal compounds. They are incompatible with acids; mixing nitriles with strong oxidizing acids can lead to extremely violent reactions. Nitriles are generally incompatible with other oxidizing agents such as peroxides and epoxides. The combination of bases and nitriles can produce hydrogen cyanide. Nitriles are hydrolyzed in both aqueous acid and base to give carboxylic acids (or salts of carboxylic acids). These reactions generate heat. Peroxides convert nitriles to amides. Nitriles can react vigorously with reducing agents. Acetonitrile and propionitrile are soluble in water, but nitriles higher than propionitrile have low aqueous solubility. They are also insoluble in aqueous acids
Chemical Properties
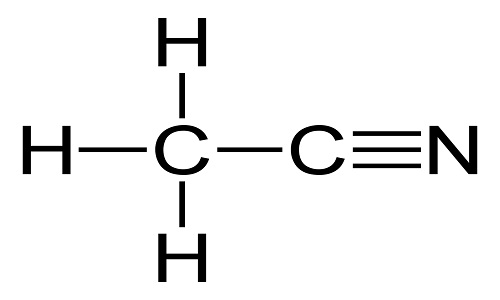
Acetonitrile (methyl cyanide), CH3CN, is a colorless liquid with a sweet, ethereal odor. It is completely miscible with water and its high dielectric strength and dipole moment make it an excellent solvent for both inorganic and organic compounds including polymers.
Chemical Properties
Acetonitrile is a colorless liquid with an ether-like odor and a polar solvent. It is the simplest
organic nitrile and is widely used. It is a by-product of the manufacture of acrylonitrile, and
acetonitrile has, in fact, replaced acrylonitrile. It is used as a starting material for the produc-
tion of acetophenone, alpha-naphthalenacetic acid, thiamine, and acetamidine. It has been
used as a solvent and in making pesticides, pharmaceuticals, batteries, and rubber products,
and formulations for nail polish remover, despite its low but signifi
cant toxicity. Acetonitrile
has been banned in cosmetic products in the European Economic Area (EEA) since early
2000 and acetone and ethyl are often preferred as safer for domestic use. Acetonitrile has
a number of uses, primarily as an extraction solvent for butadiene; as a chemical interme-
diate in pesticide manufacturing; as a solvent for both inorganic and organic compounds;
to remove tars, phenols, and coloring matter from petroleum hydrocarbons not soluble in
acetonitrile; in the production of acrylic fi
bers; in pharmaceuticals, perfumes, nitrile rubber,
and acrylonitrile-butadiene-styrene (ABS) resins; in high-performance liquid and gas chro-
matographic analysis; and in extraction and refi
ning of copper.
Waste Disposal
Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform with EPA regulations governing storage, transportation, treatment, and waste disposal. Incineration with nitrogen oxide removal from effluent gases by scrubbers or incinerators
Physical properties
Colorless liquid with an ether-like or pungent odor of vinegar. A detection odor threshold
concentration of 1,950 mg/m3 (1,161 ppmv) was experimentally determined by Dravnieks (1974).
An odor threshold concentration of 13 ppmv was reported by Nagata and Takeuchi (1990).
Application
Acetonitrile is used as a solvent for polymers, spinning fibers, casting and molding plastics, and HPLC analyses; for extraction of butadiene and other olefins from hydrocarbon streams; in dyeing and coating textiles; and as a stabilizer for chlorinated solvents. It occurs in coal tar and forms as a by-product when acrylonitrile is made. Although acetonitrile is one of the more stable nitriles, it undergoes typical nitrile reactions and is used to produce many types of nitrogencontaining compounds.Acetonitrile also is used as a catalyst and as an ingredient in transitionmetal complex catalysts.
Definition
ChEBI: Acetonitrile is a nitrile that is hydrogen cyanide in which the hydrogen has been replaced by a methyl group. It has a role as a polar aprotic solvent and an EC 3.5.1.4 (amidase) inhibitor. It is an aliphatic nitrile and a volatile organic compound.
Production Methods
Acetonitrile is mainly prepared by dehydration of acetamide (CH3CONH2) with
glacial acetic acid (Turner 1950) or by reacting acetic acid with ammonia at
400-500°C in the presence of a dehydration catalyst (Anon 1978).
Fire Hazard
Flammable liquid; flash point (open cup) 5.5°C (42°F); vapor pressure 73 torr at 20°C (68°F); vapor density at 38°C (100°F) 1.1 (air = 1); the vapor is heavier than air and can travel some distance to a source of ignition and flash back; ignition temperature 524°C (975°F); fire-extinguishing agent: dry chemical, CO2, or “alcohol” foam; use a water spray to flush and dilute the spill and keep fire-exposed containers cool.
Muraki et al. (2001) have reported a case of systemic rhabdomyolysis and acute renal failure in a 35-year old man after acetonitrile exposure. The symptoms were vomiting, convulsion, and loss of consciousness 15 hours after exposure. Initial therapy against cyanide poisoning was only partially effective.
Acetonitrile vapors form an explosive mixture with air; the LEL and UEL values are 4.4% and 16.0% by volume of air, respectively. It reacts with strong oxidizers and acids, liberating heat along with pressure increase. Thus contact in a close container can result in rupture of the container. Erbium perchlorate tetrasolvated with acetonitrile when dried to disolvate exploded violently on light friction (Wolsey 1973). Neodymium perchlorate showed similar heat and shock sensitivity when dried down to lower levels of solvation (Chemical & Engineering News, Dec. 5, 1983). Bretherick (1990) proposed that the tendency for oxygen balance to shift toward zero for maximum energy release, with diminishing solvent content, decreased the stability of solvated metal perchlorates at lower levels of solvation. Such a zero balance for maximum exotherm should occur at 2.18 mol of acetonitrile solvated to metal perchlorate. Metals such as lithium react exothermically with acetonitrile at ambient temperature (Dey and Holmes 1979).
Flammability and Explosibility
Acetonitrile is a flammable liquid (NFPA rating = 3), and its vapor can travel a
considerable distance to an ignition source and "flash back." Acetonitrile vapor
forms explosive mixtures with air at concentrations of 4 to 16% (by volume).
Hazardous gases produced in a fire include hydrogen cyanide, carbon monoxide,
carbon dioxide, and oxides of nitrogen. Carbon dioxide or dry chemical
extinguishers should be used for acetonitrile fires.
Industrial uses
Acetonitrile is used as a solvent both in industry and in the laboratory, as a
rodenticide, and in the denaturation of alcohol. Because of both its solvent
properties and volatility, it is useful for extracting vegetable and animal oils and
dissolving hydrocarbons, oils, and greases. Acetonitrile is used for the purification
of acetylene and artificial textile fibers, and as an antioxidant for rubber (Dequidt
et al 1974). It has also been used to extract herbicide residues from soils (Smith
1980), to remove tars and other compounds from petroleum hydrocarbons, and to
extract fatty acids from vegetable and fish liver oil. Acetonitrile is now a standard
solvent component in reversed-phase high-performance liquid chromatography. It
is the starting point for the syntheses of a number of organic compounds such as
carboxylic acids and various nitrogen derivatives (Smiley 1981).
Carcinogenicity
Under the conditions of these 2-
year inhalation studies by NTP, there was equivocal evidence
of carcinogenic activity of acetonitrile in male F344/N rats
based on marginally increased incidences of hepatocellular
adenoma and carcinoma. There was no evidence of carcinogenic
activity of acetonitrile in female F344/N rats exposed
to 100, 200, or 400 ppm. There was no evidence of carcinogenic
activity of acetonitrile in male or female B6C3F1 mice
exposed to 50, 100, or 200 ppm. Exposure to acetonitrile by
inhalation resulted in increased incidences of hepatic basophilic
foci in male rats and of squamous hyperplasia of the
forestomach in male and female mice.
Environmental Fate
Biological. Resting cell suspensions of the soil methylotroph Methylosinus trichosporium OB-
3b rapidly metabolized acetonitrile via oxygen insertion into the C-H bond generating the
intermediate formaldehyde cyanohydrin. The latter compound loses hydrogen cyanide yielding
formaldehyde which is then oxidized to formate (HCO2H) and bicarbonate ion (Castro et al.,
1996).
Photolytic. A rate constant of 4.94 x 10-14 cm3/molecule?sec at 24 °C was reported for the vaporphase
reaction of acetonitrile and OH radicals in air (Harris et al., 1981). Reported rate constants
for the reaction of acetonitrile and OH radicals in the atmosphere and in water are 1.90 x 10-14 and 3.70 x 10-14 cm3/molecule?sec, respectively (Kurylo and Knable, 1984). The estimated lifetime of
acetonitrile in the atmosphere is estimated to range from 6 to 17 months (Arijs and Brasseur,
1986).
Chemical/Physical. The estimated hydrolysis half-life of acetonitrile at 25 °C and pH 7 is
>150,000 yr (Ellington et al., 1988). No measurable hydrolysis was observed at 85 °C at pH
values 3.26 and 6.99. At 66.0 °C (pH 10.42) and 85.5 °C (pH 10.13), the hydrolysis half-lives
based on first-order rate constants were 32.2 and 5.5 d, respectively (Ellington et al., 1987). The
presence of hydroxide or hydronium ions facilitates hydrolysis transforming acetonitrile to the
intermediate acetamide which undergoes hydrolysis forming acetic acid and ammonia (Kollig,
1993). Acetic acid and ammonia formed react quickly forming ammonium acetate.
At an influent concentration of 1,000 mg/L, treatment with GAC resulted in an effluent
concentration of 28 mg/L. The adsorbability of the carbon used was 194 mg/g carbon (Guisti et
al., 1974).
Burns with a luminous flame (Windholz et al., 1983), releasing toxic fumes of hydrogen
cyanide.
Metabolism
Acetonitrile metabolism in dogs was demonstrated by Lang (1894), who reported
that about 20% of the nitrile administered was converted to thio-cyanate in the
urine, while guinea pigs metabolized acetonitrile to a greater extent (50% of dose
excreted as thiocyanate). When the animals were pre-treated with ethanol, acetonitrile
metabolism was induced (Tanii and Hashimoto 1986). In rats, acetone was
found to potentiate acetonitrile toxicity and elevate cyanide concentrations in the
blood (Freeman and Hays 1985). Baumann et al (1933) found that rabbits injected with acetonitrile excreted 27-35% of the dose as thiocyanate, while in thyroidectomized
rabbits, the excretion decreased significantly (3-5% of the dose).
Thiocyanate excretion was increased notably upon feeding dessicated thyroid to
these animals. Hunt (1923) found that powdered sheep thyroid protected mice
against acetonitrile toxicity. However, the role played by the thyroid in the
detoxication of cyanide to thiocyanate is unclear. It has been suggested that the
thyroid may have a role in the microsomal cleavage of cyanide from acetonitrile
other than its direct effect on sulphation of cyanide to thiocyanate.
The nature of oxidizing enzymes for nitriles in general, including acetonitrile have
been studied by Ahmed and Patel (1979). The enzymes were localized in the
hepatic microsomal fraction and required NADPH as a cofactor in the presence of
oxygen. In recent studies on the mammalian metabolism of acetonitrile, the
mechanisms of cyanide liberation, and the enzymes involved, have also been
reported by Tanii and Hashimoto (1984, 1986) and Freeman and Hays (1988).
These studies confirmed the role of microsomal mixed function oxidase in the
metabolism of acetonitrile.
Firmin and Gray (1976) studied the fate of acetonitrile in the bacterium
Pseudomonas. They found that [14C]-acetonitrile is metabolized to citrate, succinate,
fumarate, malate, glutamate, pyrrolidonecarboxylic acid, and asparate. They
reported that this species of bacteria metabolized acetonitrile by direct hydrolysis
of the cyanide moiety to acetamide. Although it is possible that a similar reaction
may occur in mammalian systems, it has not yet been reported.
storage
Acetonitrile should be
used only in areas free of ignition sources, and quantities greater than 1 liter should
be stored in tightly sealed metal containers in areas separate from oxidizers.
Toxicity evaluation
If released to ambient air, acetonitrile will remain in the vapor
phase where it will be degraded through reaction with photochemically
produced hydroxyl radicals. The half-life of acetonitrile
in ambient air has been estimated to be about 620 days. If released
to soil, acetonitrile is expected to volatilize rapidly. Biodegradation
in soil is not expected to be a major degradation pathway. If
released to water, acetonitrile is not likely to adsorb to soil and
sediment particles. Acetonitrile is expected to be removed from
water bodies through volatilization, as the chemical hydrolysis
and bioaccumulation potential for this chemical are low.