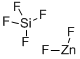Chemical Properties
White crystals.Decomposes
on heating; soluble in water.
General Description
A solid material, or the solid mixed with water. Insoluble in water and denser than water. Contact with the material may cause irritation to skin, eyes, and mucous membranes. ZINC FLUOROSILICATE(16871-71-9) may be toxic by ingestion.
Reactivity Profile
ZINC FLUOROSILICATE has weak oxidizing or reducing powers. Redox reactions can however still occur. The majority of compounds in this class are slightly soluble or insoluble in water. If soluble in water, then the solutions are usually neither strongly acidic nor strongly basic. These compounds are not water-reactive.
Air & Water Reactions
Insoluble in water.
Health Hazard
Inhalation of dust irritates nose and throat; excessive inhalation may cause severe pulmonary inflammation. Ingestion causes nausea, cramps, vomiting, shock, convulsions, cyanosis, and other symptoms of fluoride poisoning. Contact with eyes or skin causes irritation; skin ulcers may develop.
Fire Hazard
Special Hazards of Combustion Products: Toxic and irritating hydrogen fluoride and silicon tetrafluoride are formed in fires.