Hazard
As for caustic soda, ethanol. Sodium
hydroxide is formed when sodium ethylate is
exposed to moisture.
Chemical Properties
White powder, sometimes having
brownish tinge; readily hydrolyzes to alcohol and
sodium hydroxide.
Definition
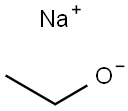
ChEBI: An organic monosodium salt that has ethoxide as the counterion.
General Description
Sodium ethoxide is an alkoxide salt mainly used as a strong base in organic reactions such as deprotonation, dehydration and dehalogenation.
Flammability and Explosibility
Highlyflammable
Purification Methods
It is a hygroscopic powder which should be stored under N2 in a cool place. A likely impurity is EtOH which can be removed by warming at 60-80o under high vacuum. It is hydrolysed by H2O to yield NaOH and EtOH. Other impurities, if kept in air for long periods are NaOH and Na2CO3. In this case the powder cannot be used if these impurities affect the reactivity, and a fresh sample should be acquired [IR: Seubold J Org Chem 21 156 1956]. [Beilstein 1 H 311, 1 IV 1289.]