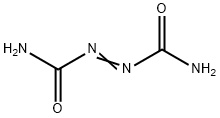
General Description
A yellow to orange powder. Insoluble in water and common solvents. Soluble in dimethyl sulfoxide. Nontoxic.
Reactivity Profile
AZODICARBONAMIDE(123-77-3) is easily ignited and burns rapidly. Confined samples show a high rate of pressure rise during thermal decomposition, which produces carbon monoxide and nitrogen. Sensitive to temperatures exceeding 122°F. May be sensitive to exposure to light. Stable in bulk when stored for two weeks at temperatures up to 140°F. Slightly unstable in water suspension (showed1.3% decomposition at 2 mg/mL over a two-week period at room temperature in the light but no decomposition at 41°F over a two-week period in the dark . Reacts with hot water to give nitrogen, carbon monoxide, and ammonia [Hawley]. Decomposes in hot hydrochloric acid. Incompatible with strong acids and bases, and with compounds of metals.
Air & Water Reactions
Highly flammable. Water insoluble. Dust may form an explosive mixture in air.
Health Hazard
Inhalation or contact with vapors, substance or decomposition products may cause severe injury or death. May produce irritating, toxic and/or corrosive gases. Runoff from fire control may cause pollution.
Fire Hazard
Self-decomposition or self-ignition may be triggered by heat, chemical reaction, friction or impact. May be ignited by heat, sparks or flames. Some may decompose explosively when heated or involved in a fire. May burn violently. Decomposition may be self-accelerating and produce large amounts of gases. Vapors or dust may form explosive mixtures with air.
Chemical Properties
orange crystalline powder
Definition
ChEBI: Azodicarbonamide is an organic molecular entity.
Flammability and Explosibility
Nonflammable